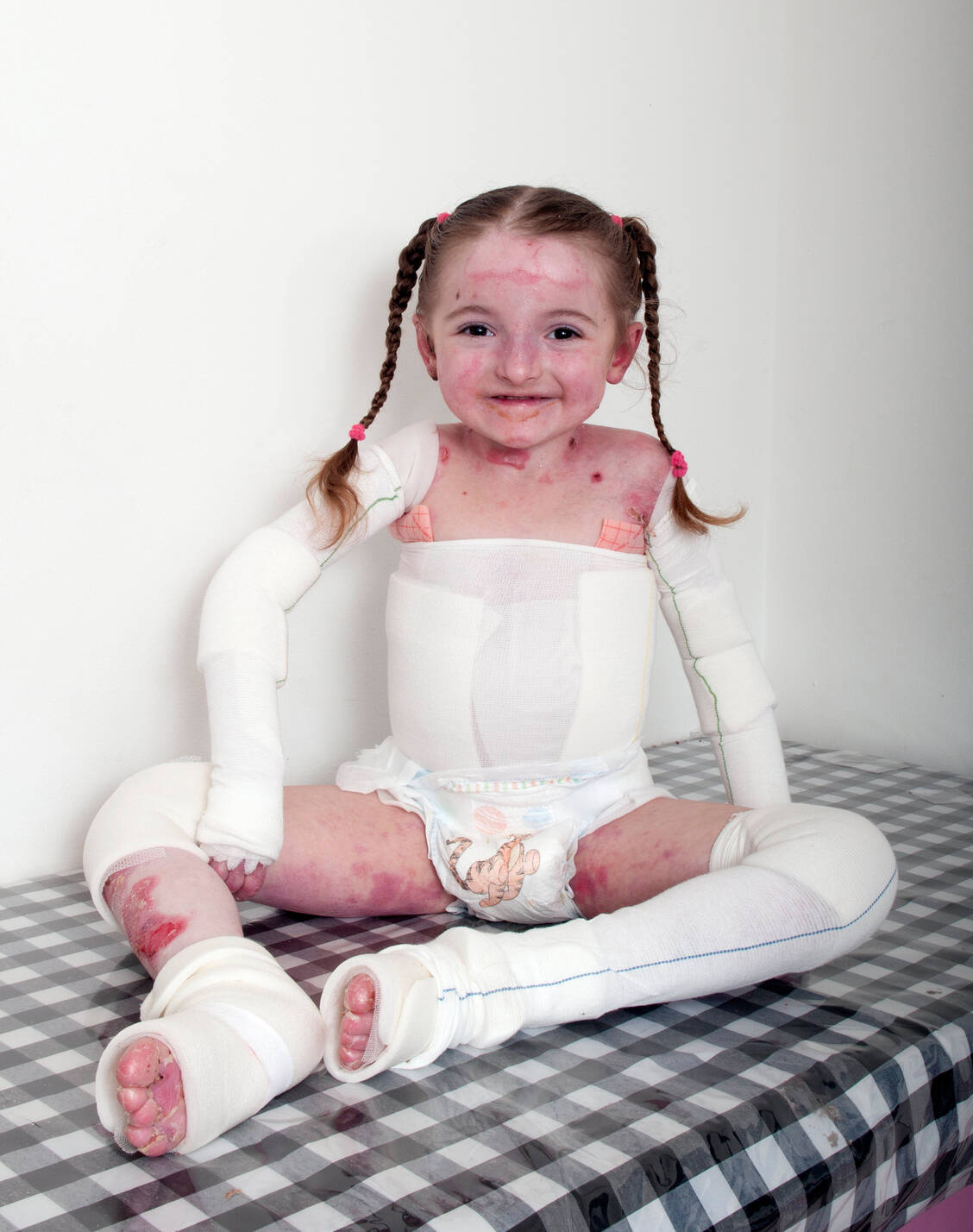
Filsuvez Significantly Reduces DEB Wounds After 15 Months in EASE Trial | Patients on Filsuvez Report Less Pain During Wound Dressings in Trial | Epidermolysis Bullosa News

Topical Morphine Gel Can Help Ease Pain of EB Wounds: Report | Gel Used to Treat 3 Patients in UK With Good Results | Epidermolysis Bullosa News

Debra Ireland - The Butterfly Skin Charity - The first medication specifically developed to treat EB has successfully completed a Phase 3 clinical trial. Filsuvez is a topical therapeutic gel developed by

Filsuvez Significantly Reduces DEB Wounds After 15 Months in EASE Trial | Patients on Filsuvez Report Less Pain During Wound Dressings in Trial | Epidermolysis Bullosa News

Filsuvez Significantly Reduces DEB Wounds After 15 Months in EASE Trial | Patients on Filsuvez Report Less Pain During Wound Dressings in Trial | Epidermolysis Bullosa News